Virtuoso Forms is a digital solution for completing health documentation that streamlines the process of collecting information.
Forms can be used to capture information directly from patients at their convenience and makes it easy for clinicians to capture data using a mobile device.



Examples of how Forms can be utilised include online assessments pre-surgery to avoid unnecessary hospital visits and PREMs and PROMs feedback for continuous service improvement. Forms also provides an innovative, mobile-optimised solution to manage clinical documentation for care teams, enabling the easy capturing of data at a patient’s bedside or out in the community.


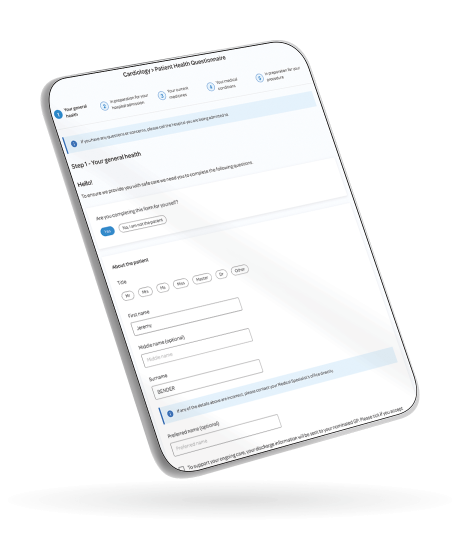

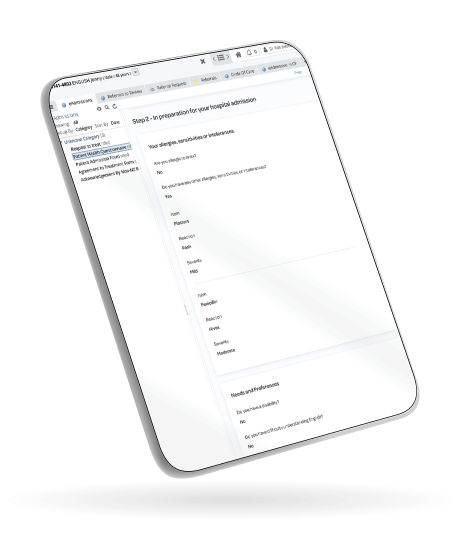
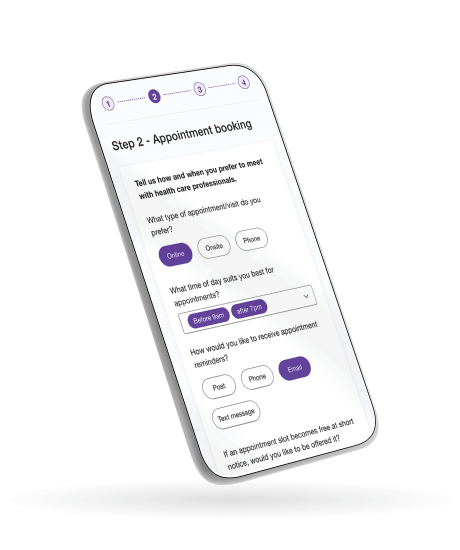
Perfectly fits to any screen size to optimise data-capturing for everyone; for example, clinicians capturing health data at the bedside or patients completing surveys on their smartphone or tablet.
Provides a flexible layout and wide range of input components and data displays. Use pre-built standard templates or build your own; for example check box or drop down question types, dynamic show/hide rules tailored to the respondent, field validations.
Forms can be easily customised to provide a personalised experience to end users aligned with customer branding.
Provides management features including different view modes depending on current document status or access (edit, read-only and view mode) and standard form actions (save draft, edit, delete, complete and cancel). Version history is also captured for clinical reference and auditing requirements.
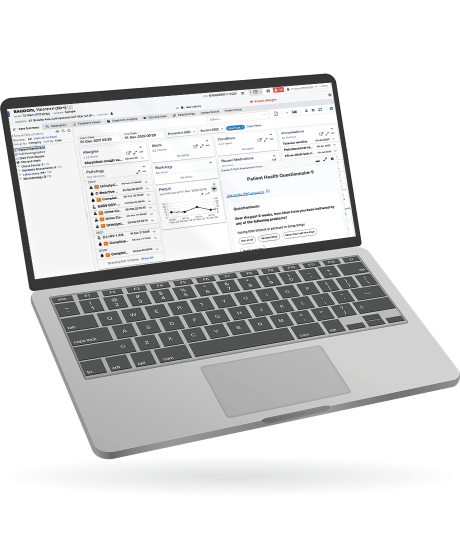
Forms has been developed with team collaboration in mind by linking patient and clinical data. Data submitted via Forms is accessible in real-time within the clinical record so that care teams are always working with the most up-to-date information.
Standalone Forms can be sent to patients via an email link to complete or Forms can be made available to patients within Patient Portal where they can perform all required actions. Forms can also be used within Clinical Portal for clinicians to capture health data and manage their clinical documentation.